
 |
Home | Store | Blog | Forums | FAQs | Lesson Plans | Pictures |
|
#1
|
|||
|
|||
|
Hi, can't buy hydroponics where I live and the shipping costs are way to much (pretty much have to pay the price of the solution in shipping costs), so I thought I will try and make my own. Information on the web is pretty scratchy so I am wondering if anyone has a recipe that they would like to share. Thanks.
|
|
#2
|
|||
|
|||
|
can view this Achiltibuie Garden - hydroponic systems
|
|
#3
|
|||
|
|||
|
Quote:
|
|
#4
|
|||
|
|||
|
Tell me what you need and I'll make you the recipe with single standard ingredients.
|
|
#6
|
|||
|
|||
|
Usually with tomatoes (unlike with other plants) vegetative vs. flowering formulas are not used as such. This is because tomatoes require high Potassium levels and low nitrogen levels all along the growing states, to prevent too much vegetative growth. Some strategies go with very high concentrate formulas, especially for huge plants with high yields. I do prefer a less aggressive strategy and have tested and used it successfully for some time. I've in fact adopted a nutrient strategy from the University of Florida and have modified it (actually simplified from 5 step) to a 2 step strategy.
Do you know how to proceed with 2 component mixes and will you manage to dissolve all salts and components properly and safely in hot water? What about micro nutrients, do you own a scale that can handle weights of 0.1 gr? In case you know how to handle it anyway, I'll just give you the recipe(s) without any detailed instructions. In case you have no experience, I'd rather give you a few hints... Is a recipe for 1000 Liter nutrient solution (some 4-5 Liter of each concentrate, A, and B) and using about 2.5 kg of raw materials a good target? Cheers, Luches Last edited by Luches; 10-21-2009 at 01:49 AM. |
|
#7
|
|||
|
|||
|
Thanks Luches, I did a bit of chemistry at university so I shouldn't have to many problems mixing the salts. As for the weights, I am about to buy some pocket scales from ebay to measure the micronutrients, was going to get .01g up to 100g and just use electronic kitchen scales for the macro nutrients.
Thanks yeah 5 litres of each part should be plenty. |
|
#8
|
|||
|
|||
|
Ok, hang on there, I'll put something together until tomorrow!

|
|
#9
|
|||
|
|||
|
OK, I've got it ready in time.
First graphics shows the chart of the 5 step program of the University of Florida, based on their research that tomato (in hydroponic) don't need more than 150 ppm of nitrogen at any state of growth. 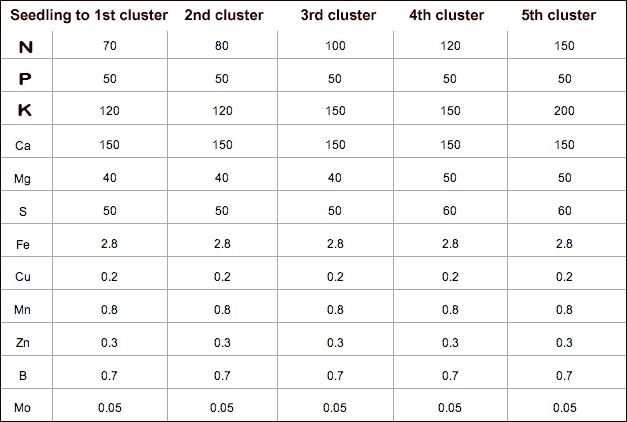 And here the RECIPE for the formula of the last stage (for 1000 Liter of final solution) which I actually used successfully. That is as close as you could possibly get "math-wise" without using lots of decimals. But this is not all you need - you probably need a starter formula that you can use until first or even second cluster. But here I need your opinion and cooperation about how you want to proceed. I can calculate any recipe for one of the 5 step formulas and even intermediate formulas. You name it I get you the recipe!   Recipe for 1000 Liter solution or 2 x 5 = 10 Liter Concentrate All measures of components and ingredients in GRAMM Please note that this is a rather conservative and low concentration formula at 1.7 EC. There are many other formulas that go much more aggressive! With this formula you need a big enough reservoir or closely monitor EC for sufficiently high concentration and/or top up and change nutrients frequently. Nothing to worry about though, only because it is a economy strategy, it requires good monitoring. PS: I've increased the Phosporus (P) slightly. This is because if used in lower concentration, you still have enough of it. A little more on the other hand (as in this formula) doesn't do any harm! In case you prefer a more "generous" formula, you tell me - I'll modify it in no time... but to reach higher K-rate, you'll need to buy one more raw material called Potassium Sulphate. Last edited by Luches; 10-22-2009 at 03:46 AM. |
|
#10
|
|||
|
|||
|
Excellent thanks Luches! I notice you have 0 for both Potassium Sulphate and Diammonium Phosphate, that is if want to increase the Potassim and Phosphate i gather, what about Nitrogen what whould you use if want to increase the nitrogen levels but not calcium?
I will just used this formular for my first grow and see how it goes from there. |
|
#11
|
|||
|
|||
|
Hi again,
Quote:
Quote:
Quote:
Best of luck an cheers! Luches |
|
#12
|
|||
|
|||
|
Quote:
Thank you |
|
#13
|
|||
|
|||
|
Quote:
 Nutrient making is quite an extensive and specific topic, even chemists get confused with it... sometimes. You should wrap your head around it by reading some online instructions and get basic knowledge, as NPK content of common components and how they are used. |
|
#14
|
|||
|
|||
|
Hello Luches. Thank you very much for the valuable information.
Bellow is the way I'm calculating the NPK Recipe for the example of the University of Florida. I write this in order you please to guide me in my calculations N: 2*14.01*666/236.17 [Ca(NO3)2*4H2O] + 14.01*331/101.1 [KNO3] = 124.88 ppm vs. 150 ppm you calculated P: 30.97*250/136.09 [KH2PO4] = 56.89 ppm vs. 57 ppm you calculated K: 39.1*331/101.1 [KNO3] + 39.1*250/136.09 [KH2PO4] = 199.84 ppm vs 200 ppm you calculated Mg: 24.305*505/246.51 [MgSO4*7H2O] = 49.79 ppm vs 50 ppm you calculated Ca: 40.078*94/219.08 [CaCl2*6H2O] = 17.19 ppm vs 15 ppm you calculated Cl: 2*35.453*94/219.08 [CaCl2*6H2O] = 30.42 ppm vs 60 ppm you calculated Could you plese help me to improve my calculations? My best. |
|
#15
|
|||
|
|||
|
Any idea of how much the savings are making you own nutrient mix?
|
|
#16
|
||||
|
||||
|
Quote:
First of all, components cannot be seen as 99.9% pure lab grade chemicals but they actually are of so called fertilizer grade quality. The content of elements may vary slightly or sometimes widely. The important thing here is: know your base material and adopt the maths to them. Example: Quote:
And around 25-30 more ppm of Calcium is provide by CaCl (at least in my formula)! Quote:
 Quote:
As for now, either way it's not an issue as only minimal quantities are required and it's actually better to have only 30 ppm of Cl than 60. An error of that range with that quantity isn't actually modifying the formula - here it actually improves it! I guess that explains it. Your maths are all right, but we have got different base products in some case(s). PS: as for the savings, Wade, it is a HUGE difference once you are break even with your equipment! Last edited by Luches; 04-07-2010 at 10:59 PM. |
 |
| Bookmarks |
| Tags |
| nutrients |
|
|